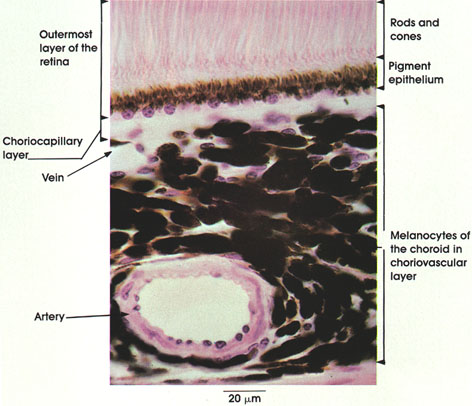
Section 5: Muscular Tissue
Muscle fibers are elongated cells with distinctive shapes specialized for shortening or contraction. These contractile fibers provide the means of movement for minute body hairs, air in respiration, ingested food and liquid, reproductive cells, blood and lymph, and small and large parts of the body. Muscle permits appropriate responses to external and internal stimuli as well as every form of communication by the individual with the external environment.
Muscular contractions, which may be coarse or extremely refined and are graded between fast and slow, are controlled by the nervous system, which is devoted in large measure to these essential activities. Muscle fibers in vertebrates may be classified structurally as nonstriated, plain, or smooth; striated cardiac; or striated skeletal. A broadened classification, which considers function, follows: smooth, involuntary; striated cardiac, involuntary; and striated skeletal, voluntary. The structural/functional classification indicates whether the contractile activity is under intentional or autonomic control.
Another functional consideration is concerned with the ability of smooth and cardiac muscle to contract spontaneously in the absence of a nerve supply (myogenic contraction). The contractile activity of involuntary muscle is normally regulated by the autonomic (sympathetic and parasympathetic) nervous system. Striated skeletal muscle fibers are totally dependent upon the nervous system, however, for both their structural integrity and function. Each striated skeletal muscle fiber is supplied with a nerve fiber ending on a specialized region of the cell membrane or sarcolemma, the subneural region of the motor end plate. If the nerve supply to a skeletal muscle is interrupted, the component muscle fibers will atrophy rapidly (denervation atrophy). If muscle is worked, it increases in size and strength; if it is not used, it will also atrophy (disuse atrophy).
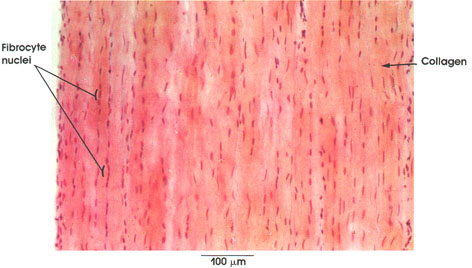
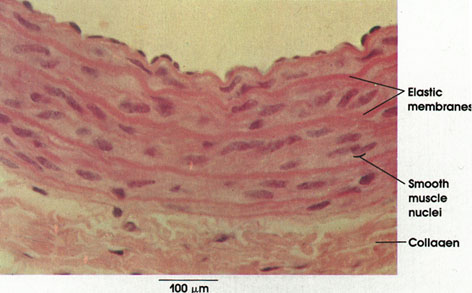
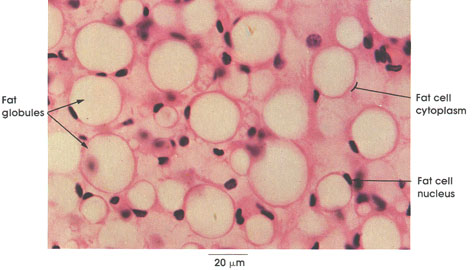
Smooth muscle fibers are generally small fusiform fibers that vary from about 15 to 200 µm in length and from 3 to 10 µm in diameter. Each muscle fiber possesses a single, elongated nucleus, which characteristically becomes shorter and broader and may coil when the muscle fiber contracts. Smooth muscle fibers may occur singly, as in the scrotum (tunica dartos); in small bundles or fascicles associated with hair follicles (arrector pili muscle); in well-defined, layered sheets that are coiled, as in muscular arteries, or arranged in two thick layers at right angles to each other, as in the intestines; or with an additional layer in an irregular pattern, as in the stomach, bladder, and uterus. Branched smooth muscle fibers can be found in the nipple of the mammary glands and in the enclocardium of the atrium of the heart.
Smooth muscle fibers usually contract slowly but are capable of sustained contractile activity. Most of the smooth muscle fibers of the gastrointestinal and genitourinary tracts are linked to each other by specialized surface membrane (sarcolemmal) contacts (gap junctions or nexus), which transmit electrical excitatory stimuli from cell to cell. The gap junction can be seen in the introductory plates of this book. This structural/functional arrangement permits large numbers of smooth muscle fibers to be activated sequentially by a minimal nerve supply. The excitatory nerve impulse is transmitted to a smooth muscle fiber, which conducts it over its surface, across the gap junction to another fiber, which passes it on, resulting thereby in a sustained and coordinated contraction (peristalsis) over long distances.
Cardiac muscle fibers are generally larger than smooth muscle fibers and appear cross-striated when stained or examined with polarized light. Cardiac muscle fibers are joined serially end to end and characteristically branch to unite with adjacent fibers. Cardiac muscle fibers form a functional but not a protoplasmic syncytium. The junctional site between fibers is called the intercalated disc. The intercalated disc is composed of two important components: the adhesion plate (desmosome) between adjacent cells and the gap junction (or nexus), which allows the electrical excitatory impulse to be transmitted from cell to cell in the same way as in smooth muscle, resulting in a synchronized coordinated contraction relaxation cycle essential to normal heart function. The branched cardiac fibers possess one or two nuclei, which are centrally located. The contractile substance of the cardiac fiber is organized into subunits called myofibrils, which are cross-striated. The cross striations will be discussed in relation to striated skeletal muscle. The myofibril characteristic of cardiac and skeletal muscle is not seen in smooth muscle, although the myofilaments of which the myofibril is composed are found in all three muscle fiber types. Myofilaments cannot be resolved by the light microscope, although Brücke (1858) postulated their existence based on polarization microscopy data analysis and Kölliker (1888) suggested that the hypothetical myofilaments were composed of the newly discovered protein myosin (Kühne, 1864).
Striated skeletal muscle fibers vary in length between 2 and 25 cm, depending upon the muscle. The diameter of a single muscle fiber is also variable but is usually between 10 and 100 µm. Normal mature striated skeletal muscle fibers are irregularly polygonal in shape (Bowman, 1840), whereas developing fibers are small and round; pathologic muscle fibers tend to be round or sharply angular and usually abnormally small. The multinucleated skeletal muscle fibers, unlike smooth and cardiac muscle fibers, are not structurally or functionally uniform. Two or more distinct muscle fiber types have been identified in man and other species by light and electron microscopy, histochemistry, and functionally. Characteristically, the nuclei of skeletal muscle fibers are located peripherally adjacent to the outer limiting membrane or sarcolemma. The usual (normal) position of skeletal muscle nuclei and the discovery of the sarcolemma is credited to Bowman (1840). The nuclei of developing muscle and of cardiac muscle are typically centrally located within the muscle fiber (Bowman, 1840).
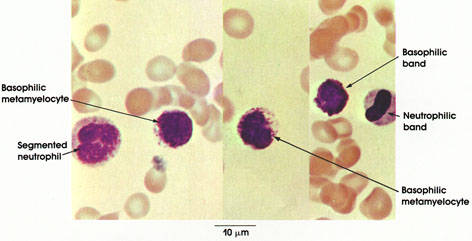
In certain skeletal muscle fibers, namely the red or slow contracting muscle fibers, the nuclei may be found scattered throughout the sarcoplasm. Based upon structural/functional studies, living muscle fibers that appear red are designated, in man, as Type I muscle fibers. These muscle fibers contain many mitochondria (Kölliker, 1857), they store and utilize lipid (droplets) metabolically, and they are red in color and contract slowly. One type of red fiber requires only a single stimulus, whereas a second type requires multiple stimuli to initiate a contraction. The former is designated a slow twitch muscle fiber, and the latter as slow tonic muscle fiber. Lorenzini (1678) first noted color differences in muscles; some are red in color, others are white. Kühne (1850) analyzed the intrafiber pigment and reported its similarity to hemoglobin. Lankester (1871) noted that although red muscles were slow in contracting, they were the most active and strongest and capable of sustained contractile activity. He also contrasted red pigeon breast muscle with white chicken breast muscle and, with both birds, capacity for sustained flight. Gunther (1921) introduced the term myoglobin for the red (intrafiber) pigment. Most whole muscles are a mixture of red and white muscle fibers, which vary in number for any particular muscle; these are seen histologically in thin sections.
Muscle fibers that appear white contain few mitochondria, store and utilize glycogen (Bernard, 1855), are essentially devoid of myoglobin, and contract rapidly but fatigue quickly. They are normally larger than red fibers. In man, these are designated as Type II muscle fibers.
In cross section, skeletal muscle fibers are seen to be composed of numerous small aggregates (1 to 2 µm) of contractile substance, the myofibrils. Myofibrils are composed of myofilaments (Hall, Jakus, Schmidt, 1946). Huxley (1954) has shown two types of myofilaments: a thick, A band myofilament 1.6 µm in length; and a thin, 1 band myofilament 1.0 µm in length, which extends from the Z line into the A band for a variable distance.
In longitudinal section, the muscle fiber and the myofibril appear cross-striated (Leeuwenhoek, 1674). The darkly stained segments, 1.6 µm in length (Krause, 1868), is designated the A band (anisotropic band), which is a region of high refractive index and is birefringent when examined with a polarizing microscope (Brücke, 1858). Alternating with the A bands is a lightly staining region of variable length, the I band (isotropic band), which is a region of low refractive index (Brücke, 1858). The I band is bisected by a thin dark-staining Z line. When a muscle fiber contracts, the band pattern changes with the A bands moving toward each other and meeting at the Z line (Bowman, 1840); the I bands disappear, and the middle of the A band becomes dark. The band pattern changes can be precisely related to the movement of myofilaments in relation to each other. Additional structural/functional details of the contractile process will be considered in the legends of the plates of this section.
STRIATED MUSCLE
Embryonic tissue
cross section
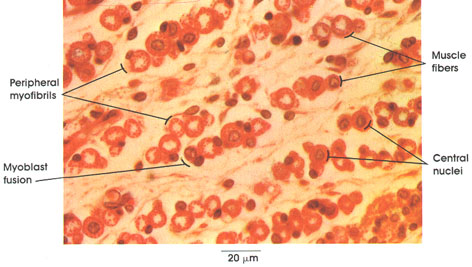
Human, Helly's fluid, H. & E., 612 x.
Developing muscle fibers are seen in different stages of development. Embryonic muscle fibers are characterized by centrally placed nuclei and peripherally disposed myofibrils. These fibers grow in length and diameter by myoblast fusion. No nuclear divisional figures are ordinarily seen at this stage. As the muscle fiber matures, the nuclei become located primarily beneath the sarcolemma at the periphery of the fiber.
A congenital disorder of skeletal muscle seen in children (centrovacuolar myopathy) is characterized by a histologic picture identical with that seen in embryonic skeletal muscle.
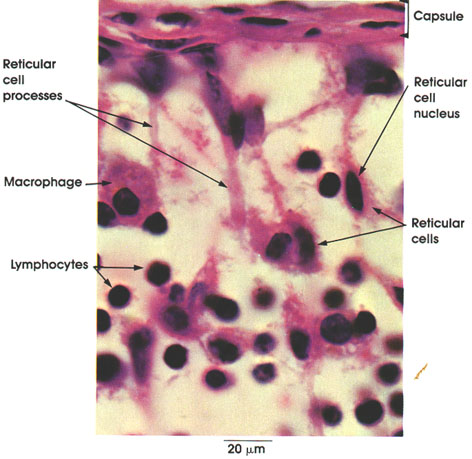
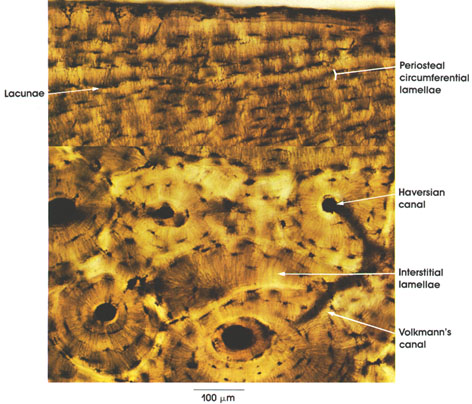
Sarcolemma
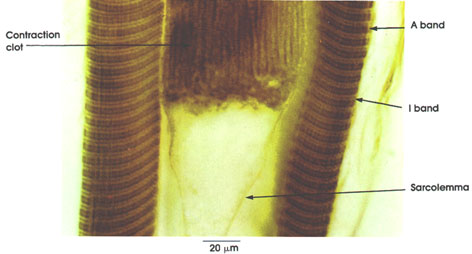
Sarcolemma: External limiting membrane of muscle fibers. Not ordinarily seen in light microscopic preparations. Seen here because of artifactual retraction of contractile elements. This artifact permitted Bowman, in 1840, to demonstrate the membrane and to name it the sarcolemma. The true sarcolemma, very much thinner than seen here, is responsible for the conduction and spread of electrical impulses from the motor end plate over the entire muscle surface, resulting in contractile activity. Electron microscopy has shown the sarcolemma to be 100 Å or 10 nm in thickness. The apparent increase in thickness rendering it visible in this preparation is due to adherent stainable sarcoplasm and, externally, to a thin basement membrane and associated reticular connective tissue fibers.
Polarization microscopy

The names given to the two major transverse striations of skeletal and cardiac muscle are derived from the studies of Brücke* (1858). With routine light microscopic techniques, alternating dark and light bands are seen within striated muscle fibers . Polarization microscopy reverses the appearance of the dark band, which becomes bright, and the light band, which appears dark. The dark band of routine light microscopy, exhibiting birefringence with polarized light, is anisotropic and is called the A band. The light band of routine light microscopy is poorly refractile and relatively isotropic and is called the I band.
Muscle fibers: Showing cross striations formed by alternating segments of high and low refractive index resulting from their submicroscopic structure, which is revealed by electron microscopy.
A band: Anisotropic band.
I band: Isotropic band. Note the birefringence or anisotropy of the Z line in the center of the I band.
Transverse striations
longitudinal section
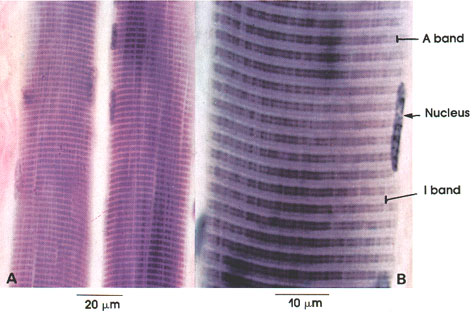
hematoxylin stain, A. 612 x; B. 1416 x.
Phosphotungstic acid hematoxylin is a stain particularly suited for the demonstration of striations in skeletal muscle. Iron hematoxylin and Mallory-azan are also effectively used for this purpose. Note that at low magnification only the two major cross striations can be seen. The dark band is the A band, and the light band is the I band. Higher magnifications are usually required to see the light-staining area in the center of the A band, which is known as the H zone, and the thin, dark line bisecting the I band, which is named the Z line. The repeating structural unit between two Z lines is called a sarcomere.
Tongue

I band: Isotropic band determined by polarization microscopy. Note the darker Z line bisecting the I band. Electron microscopy has shown that the I band contains thin filaments 50 Å or 5 nm in diameter and approximately 2 µm in length. The contractile protein actin is found in the thin I band filaments.
A band: Anisotropic band determined by polarization microscopy is bisected by the lighter H zone. The A band contains filaments of the protein myosin, which are 100 Å or 10 nm in diameter and 1.6. µm in length, as well as thin (actin) filaments extending from the I band into the A band. The A band filaments are composed of myosin molecules, which, through enzymatic activity (adenosine triphosphatase), release energy essential for the contractile process.
Z line: Bisects the I band. Z from the German word Zwischenscheibe, or in-between line.
H zone: Variable in width, it bisects the A band and vanishes in contraction as the thin I band filaments are drawn further into and through the middle of the A band. When seen, it contains only thick filaments. H for the German word Hell (bright) and also for the zone's discoverer, Hensen.*
Sarcomere: The contractile substance between two Z lines constitutes a convenient structural unit but not the precise functional or primary contractile unit of muscle fibers. See Plate 68.
Nucleus: Elongated and located beneath the sarcolemma. The sarcolemma is seen in Plate 64.
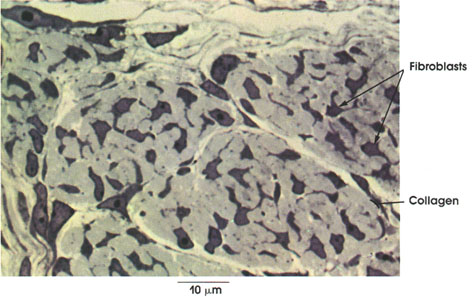
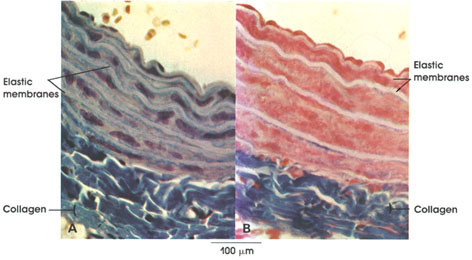
Collagen: Bundles of this connective tissue separate individual muscle fibers (endomysium), bind fascicles, or bundles, of muscle fibers (perimysium), and invest the entire muscle (epimysium). Through this tough and inelastic connective tissue, contractile forces are transmitted to bone and skin.
Relaxed and contracted
muscle fibers

A., C., phosphotungstic acid hematoxylin,
B. Mallory-azan,
D. H. & E.; 4416 x.
In this plate, the structural basis of skeletal muscle fiber contraction is shown.
A: Relaxed fiber showing distinct cross striations, the darker staining A band and the lighter staining I band. Note that the I band is bisected by a thin but deeply staining line (Z line), while the A band is bisected by a lightly staining line (H zone).
B: A fiber seen in the relaxed state except for a small segment of localized contraction. Note the change in the band pattern in this segment. Two adjacent A bands are in contact, and the I band has disappeared.
C: A fiber shown with both a relaxed and contracted segment. The A and I bands are clearly outlined in the relaxed segment but not in the contracted segment. In contraction, the I band becomes narrower and disappears. The A band does not normally become shorter except in extreme contraction. Contraction bands appear as a result of an increase in density and staining of the Z line.
D: A portion of a fully contracted muscle fiber is shown. The changes here are similar to those described in C for a contracted segment except that the normal distance between the thickened Z lines (contraction bands) is reduced, denoting extreme contraction.
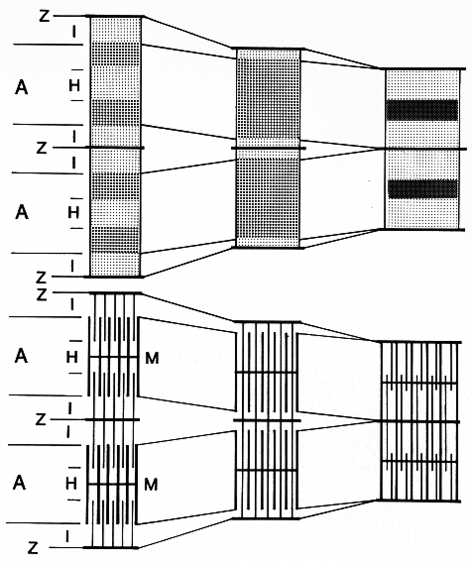
The upper figure, showing two sarcomeres, accounts for the usual light microscopic appearance (i.e., the staining densities) of sarcomeric cross striations in relaxed (left), contracting (middle), and fully contracted (right) skeletal and cardiac muscle fibers.
In the lower figure, also showing two sarcomeres, the comparable electron microscopic, ultrastructural configuration is shown. The appearance of cross striations or bands by both light and electron microscopy have their basis in the relative position and resulting density of the two major sets of myofilaments that constitute the sarcomere, that is, thin (actin) filaments, emanating from the Z line, and thick (myosin) filaments, held in hexagonal register at the M line, and their relative interdigitation in relaxation and in shortening (contraction).
Lateral rectus cross section
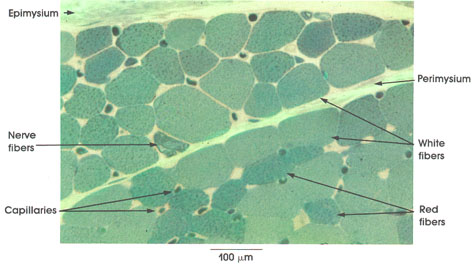
toluidine blue stain, 162 x.
Epimysium: Envelope primarily composed of collagenous connective tissue wrapping the entire muscle.
Perimysium: Connective tissue partitions between bundles, or fascicles, of muscle fibers.
White fibers (A fibers): Also known in the human as Type II fibers. These large fibers demonstrate pronounced myofibrillar ATPase activity and glycogen stores. These fibers are fast contracting.
Red fibers (B fibers): Also known in the human as Type I fibers. Characteristically smaller than white fibers, they contain numerous mitochondria and lipid stores. These fibers are slow contracting. .
Note the variation in fiber diameter. Normally, skeletal muscle fibers vary from 10 to 100 µm in diameter, depending upon muscle and species.
Nerve fibers: Somatic motor nerve fibers are distributed in the connective tissue septa of the muscle. These terminate on individual muscle fibers.
Capillaries: Widely distributed in the connective tissue septa (endomysium) between and around individual muscle fibers. Blood cells are seen within some capillaries.
Cremaster muscle myofibrils
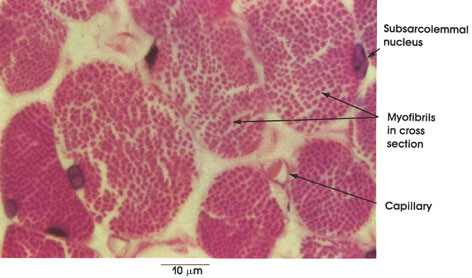
A capillary containing a red blood cell is seen between muscle fibers. Muscle fibers are provided with a rich capillary network that supplies essential nutrients and oxygen and removes metabolic wastes.
Myofibrils: Subunits of each muscle fiber. Each myofibril is composed of myofilaments. Myofibrils vary in size, depending upon the number of myofilaments they contain. Myofilaments cannot be resolved by the light microscope.
Nucleus with nucleolus: The nucleus shown in this mature muscle fiber is characteristically located near the sarcolemma. In some skeletal muscles, particularly those that are slow contracting (Type I, Type B, or red), the nuclei may be found more centrally located within the muscle fiber.
Semitendinosus, cross section
Mitochondria; succinic dehydrogenase localization

Histochemical methods similar to the one used here, which is specific for mitochrondria, have been instrumental in distinguishing muscle fiber types in health and disease.
Red muscle fiber (B fiber): Rich in mitochondria and lipids, this type of fiber is slow contracting. Known in the human as Type I muscle fiber.
White muscle fiber (A fiber): Relatively poor in mitochondria and lipids, but rich in myofibrillar ATPase activity and glycogen, these fibers are fast contracting. Known in the human as Type II muscle fiber.
Intercostal muscle
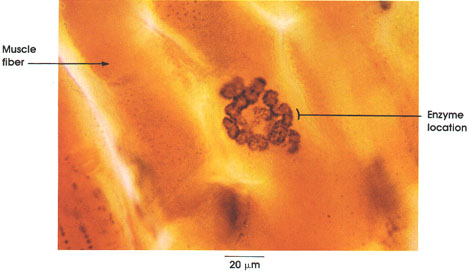
Enzyme location: The muscle sarcolemma that forms the primary and secondary clefts of the subneural apparatus is rich in acetylcholinesterase activity. Axon terminals (not seen in this preparation) lie within the primary synaptic cleft and liberate acetylcholine when a nerve action potential, originating in the spinal cord, arrives at the nerve endings. Acetylcholine results in the depolarization of the muscle membrane and the appearance of a muscle action potential, which spreads over the muscle fiber leading to muscular contraction. The acetylcholine is hydrolyzed by the enzyme located on the sarcolemma beneath the nerve terminals, and the sarcolemma is repolarized in preparation for the next nerve impulse.
Cross section
A. tongue B. sartorius
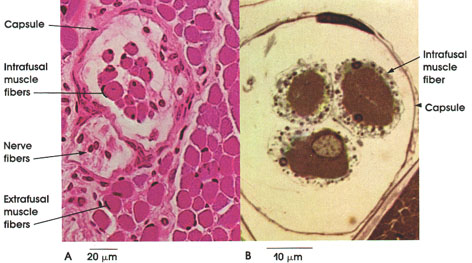
B. Frog, glutaraldehyde-osmium fixation, toluidine blue stain, 1416 x.
This plate shows some of the histological features of neuromuscular spindles as seen in the human tongue (A) and frog sartorius (B). Note that the neuromuscular spindle is surrounded by skeletal muscle fibers (extrafusal fibers). Each spindle contains several small muscle fibers (the intrafusal fibers), myelinated nerve fibers enclosed within a connective tissue capsule, which is pierced by the nerve fibers reaching the spindle. Nerve fibers of the spindle are both sensory and motor. Information conveyed from and to the muscle spindle is not consciously received but is important in reflex regulation of muscle tone. Intrafusal muscle fibers of the spindle receive axons of the gamma motor neurons in the spinal cord, whereas the extrafusal muscle fibers receive axons of the larger alpha motor neurons. .
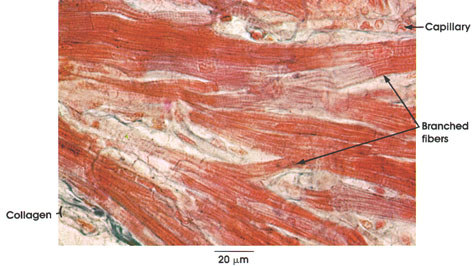
The characteristically branched cardiac muscle fibers are separated by collagenous connective tissue. The differentiation of collagenous connective tissue and cardiac muscle is clearly seen with this stain. Note the capillary containing red cells. Compare capillary diameter (approximately 8 µm) with that of the cardiac muscle fiber.
Longitudinal section
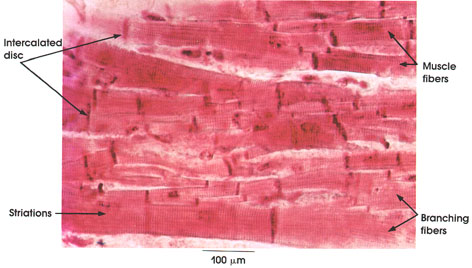
Muscle fibers: Each 9 to 22 µm in diameter, serially arranged in columns with short branches contacting adjacent fibers.
Branching fibers: Characteristic of cardiac muscle fibers. Each branching fiber limited by an intercalated disc constitutes a single muscle fiber. .
Striations: A and I bands. A bands are usually inconspicuous. Z lines are particularly prominent. Contractile apparatus and cross striations, although not usually stained well in cardiac muscle, are similar to those found in skeletal muscle (see Plates 65, 66 and 67).
Intercalated disc: Site of termination and junction of adjacent cardiac muscle fibers. Consists of snugly fit projections and indentations of adjacent cell membranes. Intercalated discs are the sites of transmission of excitatory impulses from cell to cell and provide firm attachment for contiguous fibers. The stain used in this preparation is particularly useful for demonstrating intercalated discs.
Relaxed and contracted muscle fibers
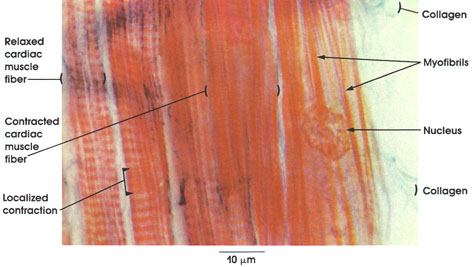
This is a longitudinal section of cardiac muscle stained with Mallory-azan, which differentiates muscular tissue (red-brown) from collagenous connective tissue (blue).
Several muscle fibers are seen. A vesicular nucleus is seen in one. The muscle fibers are separated by narrow spaces containing delicate strands of collagen fibers. Each muscle fiber is formed of subunits, the myofibrils. To the extreme left of the figure, a muscle fiber in the relaxed state is shown. Note the distinct striations. Adjacent to this fiber is another relaxed fiber except for a small area of localized contraction. Note that the muscle striations are less distinct in the contracted area. in the middle of the plate is a contracted fiber in which the striation pattern is indistinct, although the Z lines are evident.
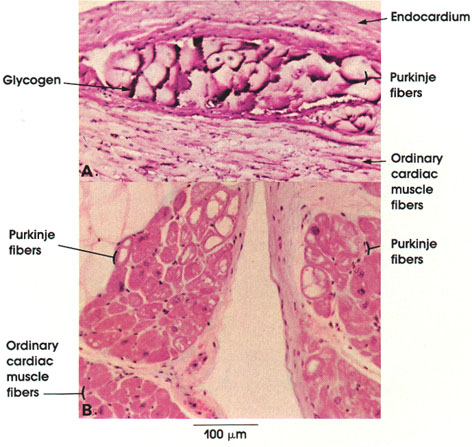
B. Human, 10% formalin, H. & E., 162 x.
In this figure, the contrast between ordinary cardiac muscle fibers and their specialized variety, the Purkinje fibers, is evident. Purkinje fibers are larger than ordinary cardiac muscle fibers and stain less intensely. Note the clear areas in the cytoplasm of Purkinje fibers in B. These represent areas from which glycogen was lost during the preparation of the tissue. By contrast, in the Purkinje fibers seen in A, the glycogen is preserved by the fixation method used. Note the subendocardial location of Purkinje fibers.
Purkinje fibers cross section

Purkinje fibers: Larger and paler than ordinary cardiac muscle fibers. Areas of clear sarcoplasm represent regions that normally contain glycogen as well as areas devoid of myofibrils. These fibers contain irregularly arranged thin myofibrils and nuclei.
Purkinje fibers cross section

Purkinje fibers: Larger and paler than ordinary cardiac muscle fibers. Areas of clear sarcoplasm represent regions that normally contain glycogen as well as areas devoid of myofibrils. These fibers contain irregularly arranged thin myofibrils and nuclei.
Duodenum longitudinal section
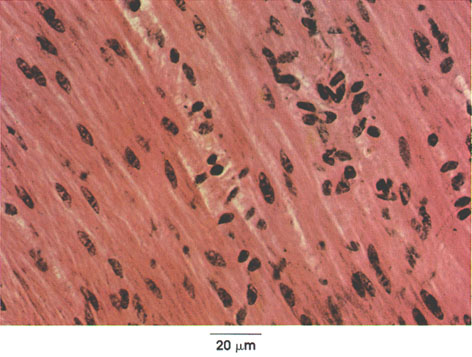
Outer longitudinal layer of smooth muscle in the tunica muscularis of the duodenum. Muscle fibers are divided by connective tissue septa into bundles. Each muscle cell has a central nucleus and abundant sarcoplasm. The muscle fibers are long, slender, and spindle-shaped. Note that differentiation between smooth muscle cells and connective tissue fibers is difficult in this preparation because of the staining method used (H. & E.). Differentiation of smooth muscle .
Duodenum cross section
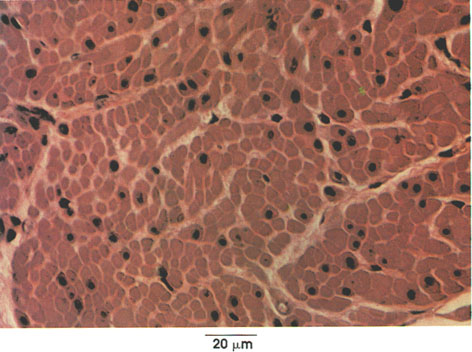
Bundles of smooth muscle fibers separated by connective tissue septa. Each fiber is characterized by abundant sarcoplasm and central nucleus. Myofibrils are not seen in smooth muscle. Note variation in cross-sectional diameter, which can be accounted for on the basis of their spindle shape (as seen in longitudinal section).
A. Longitudinal and circular (jejunum)
B. Small fascicle (skin)
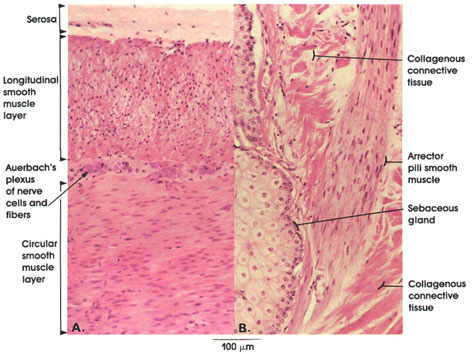
This plate shows smooth muscle fibers from two locations. In A, they are seen distributed as an outer longitudinal and an inner circular layer in the wall of the jejunum. These two layers are separated by connective tissue and by neurons and fibers of Auerbach's* autonomic plexus. In B, a smooth muscle fiber bundle of the arrector pili muscle is seen between bundles of connective tissue in the skin. Note the elongated nuclei and homogeneous cytoplasm. Arrector pili muscles originate in the papillary connective tissue and insert on hair follicles. Their contraction erects hairs in animals and produces "goose-flesh" in man. Note the proximity of the arrector pili muscle to a sebaceous gland.